
Muscarinic acetylcholine receptors (mAChRs) are a family of G protein‑coupled receptors (GPCRs). As the primary signaling receptors for acetylcholine (ACh), they are widely distributed in various human tissues, including the cardiovascular system, kidneys, gastrointestinal tract, lungs, as well as the central and peripheral nervous systems
The Muscarinic Receptor Family and Subtype Characteristics
The muscarinic receptor family comprises five subtypes, M1 to M5, serving as important drug targets for neurological disorders. They are involved in regulating multiple neural circuit functions, such as neuronal excitability, synaptic plasticity, and acetylcholine release. All subtypes can be detected in neurons and glial cells of the brain, but their expression levels vary significantly across different brain regions. This distribution specificity underlies the functional diversity of each subtype and provides a foundation for the development of subtype‑selective drugs.
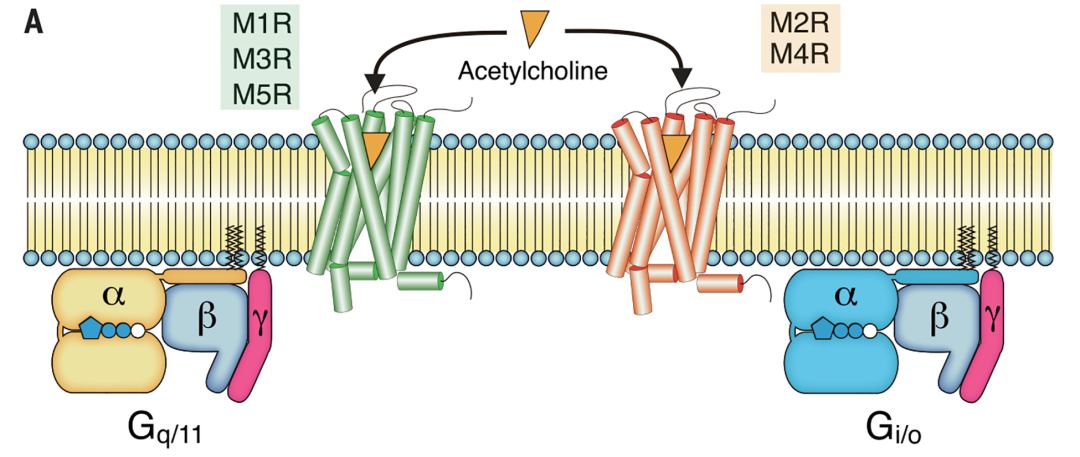
Signaling Pathways of Muscarinic Receptors
• M1: Predominantly expressed in the forebrain, cerebral cortex, hippocampus, and striatum, accounting for 60% of total muscarinic receptor expression in the central nervous system (CNS). It participates in learning and memory processes and is a key target for cognitive impairment in Alzheimer’s disease (AD).
• M2: Widely expressed in the CNS, including the basal forebrain, thalamus, and cortex. It modulates cognitive function, emotion, and behavior, and plays a vital role especially in learning and memory.
• M3: Mainly distributed in the hypothalamus, cerebral cortex, and hippocampus, with relatively low expression in the CNS. It is associated with autonomic functions such as appetite and body temperature regulation.
• M4: Predominantly located in brain regions closely linked to mental activity, including the striatum, cortex, and hippocampus. It regulates dopaminergic activity and is an important target for schizophrenia and drug addiction.
• M5: Present in various brain regions including the hippocampus, hypothalamus, and different midbrain areas, but at extremely low levels, representing less than 2% of total mAChR expression in the brain. Studies suggest that the M5 receptor is involved in the physiological regulation of cerebral arteriolar and arterial diameter.
Given their critical functions in the CNS, abnormal expression or dysfunction of muscarinic receptors is closely implicated in the pathogenesis and progression of multiple neuropsychiatric disorders, including major depressive disorder, bipolar disorder, schizophrenia, dementia, Alzheimer’s disease, and anxiety disorders.
近年来,针对毒蕈碱受体的药物开发已成为神经精神疾病领域的研究热点,并已有药物成功商业化。
In recent years, drug development targeting muscarinic receptors has become a research hotspot in the field of neuropsychiatric diseases, with several drugs successfully commercialized.
In addition, multiple drug candidates are in clinical development. For example, ML‑007C‑MA has entered Phase II clinical trials for two indications: schizophrenia and psychotic symptoms associated with Alzheimer’s disease (ADP). In December 2025, the ADP indication was granted Fast Track Designation by the U.S. FDA.CNS News | MapLight Secures $370 Million in Funding, Signaling Renewed Investor Confidence in CNS Drug Development
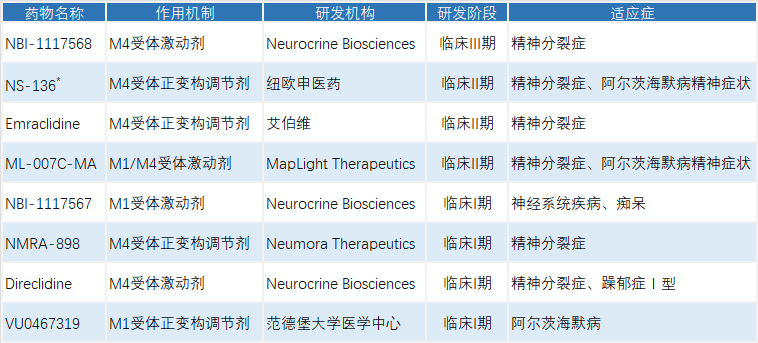
Selected Investigational Muscarinic Receptor‑Targeted Drugs and Progress
*Kylin Lab assisted NeuPharma in successfully achieving clinical translation of NS‑136 in July 2024
While the role of muscarinic receptor‑targeted drugs in schizophrenia, Alzheimer’s disease and other disorders has been validated, their development still faces several common challenges, including precise target screening, reliable evaluation of pharmacological efficacy, and verification of drug safety.
Focused on CNS Drug R&D: Building a Highly Translatable Preclinical Evaluation System
Kylin Lab specializes in preclinical services for central nervous system drug development. We have established a multimodal drug evaluation system centered on receptor and ion channel target screening, providing end‑to‑end, highly translatable technical services for innovative drug R&D:
• Target Screening Platform: Conducts precise screening for core CNS‑related targets, covering a variety of neurotransmitter receptors and key ion channels, enabling rapid detection of target activity.
• Electrophysiology Platform: Leverages a professional electrophysiology platform to accurately evaluate in vivo efficacy using objective electrophysiological indicators, providing critical data for drug development.
• Humanized Models: Based on human iPS‑derived cells and organoid models, it simulates the real human pathophysiological environment, significantly improving the success rate of clinical translation.
• Multimodal Evaluation System: Integrates behavioral assessment, biomarker detection and other technologies to achieve a full‑chain evaluation spanning target screening, functional validation, in vivo efficacy assessment and mechanistic research, offering one‑stop preclinical drug R&D services for CNS innovative drug development.

References:
Daniel J Foster, P Jeffrey Conn. Exploring novel non-opioid pathways and therapeutics for pain modulation.Neuron 2017 May 3;94(3):431-446.
Shoji Maeda, Qianhui Qu, Michael J Robertson, et al.Structures of the M1 and M2 muscarinic acetylcholine receptor/G-protein complexes.Science. 2019 May 10;364(6440):552-557.
瀚枢生物
CNSxplore | 提供一站式神经系统药物研发解决方案
Kylin Lab is a preclinical CRO company specializing in central nervous system (CNS) diseases, dedicated to offering one stop solutions for CNS drug discovery. With a portfolio of fully-validated cellular and animal disease models, combined with comprehensive research and analytical capabilities, we empower clients to accelerate the development of innovative therapies and reduce clinical trial risks.
Kylin Lab boasts an experienced technical team well-versed in international regulations, along with high-standard experimental platforms. Our core technologies include AI-driven phenotypic screening, humanized stem cells and organoids, electrophysiology, high-throughput electroencephalography (EEG), histology, and molecular biology. Our expertise spans a broad range of areas including Alzheimer’s disease, Parkinson’s disease, depression, schizophrenia, spinal muscular atrophy (SMA), pain, and stroke etc.
Since its establishment, Kylin Lab has adhered to the philosophy of “Pioneering R&D, Leading in technology Quality-first, Customer-centric.” We have served hundreds of clients, successfully completed numerous thematic research projects and IND submissions, and established a high-quality, stable, and forward-looking efficacy evaluation system.