

The 2025 China Sleep Health Survey Report shows that 48.5% of people aged 18 and above in China suffer from sleep disturbances, among which 51.1% of women are affected, higher than the 45.9% of men. Issues such as reduced energy and daytime sleepiness caused by insufficient sleep have severely impacted public quality of life. Given the limited efficacy of lifestyle interventions, the rigid demand for safe, long-term usable insomnia drugs has become prominent, making insomnia medications a highly promising blue‑ocean market.
Current Status of Clinical Insomnia Medications
The domestic clinical landscape for insomnia drugs features a layered structure: traditional drugs as the foundation, innovative drugs expanding capacity. Classic sedative‑hypnotics represented by benzodiazepines and non‑benzodiazepines remain the cornerstone of clinical use. Meanwhile, novel insomnia drugs with superior safety and more precise targets have been gradually approved and adopted clinically, driving the upgrade of insomnia treatment from forced sedation to physiological sleep regulation.
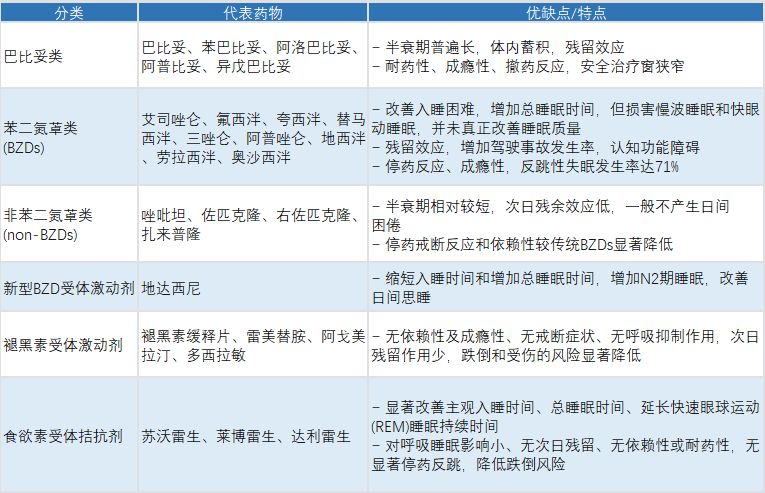
Classification and Characteristics of Common Insomnia Medications
Notably, in November 2023, China approved its first innovative insomnia drug in nearly two decades — daridorexant (Note: corrected to daridorexant as per standard international nomenclature; original text: didaxini). As a GABAAreceptor partial positive allosteric modulator acting selectively on the α1 subunit, it improves sleep with lower tolerance and dependence risks. In the first three quarters of 2025, daridorexant’s sales exceeded 100 million yuan, confirming strong market demand for safe innovative therapies.
At the same time, orexin receptor antagonists are leading a paradigm shift in insomnia treatment. Orexin is a wakefulness‑promoting neuropeptide synthesized and secreted in the lateral hypothalamus, maintaining arousal by regulating OX1 and OX2 receptors. Since suvorexant — the world’s first dual orexin receptor antagonist — was approved in 2014, lemborexant, daridorexant, and vornorexant have followed, demonstrating the tremendous clinical translation potential of this target.Related reading: Phase III results of daridorexant released; orexin receptor antagonists lead insomnia drug development
Global R&D Focus
The success of daridorexant and orexin receptor antagonists has opened a new landscape for insomnia drug development. Judging by R&D trends, orexin receptor‑targeted medicines have become the most promising innovative direction in sleep therapeutics. Multiple pharmaceutical companies are laying out this target, advancing insomnia treatment toward greater precision and safety.
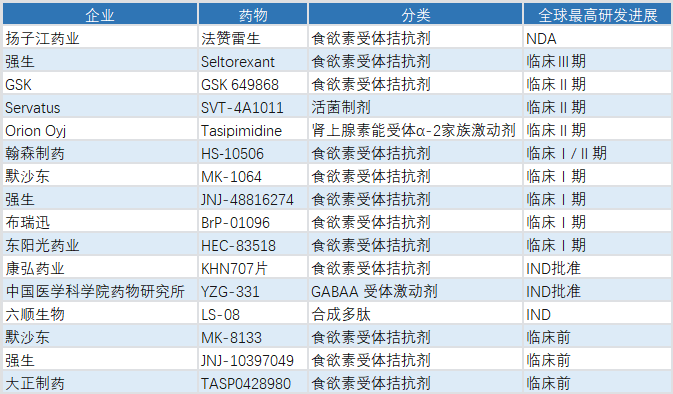
Selected Pipeline Insomnia Drugs and Progress (as of March 20, 2026)
Kylin Lab’s "Sleep Platform" Empowers Sleep Drug R&D
Kylin Lab specializes in preclinical CNS R&D services, providing comprehensive preclinical support for sleep drug development:
• Animal Models: Establish models including jet lag, postoperative pain‑related sleep disturbance, caffeine‑induced sleep deprivation, cataplexy, etc., adapting to diverse sleep disorder research scenarios.
• Technology Platform: Based on electrophysiology, continuously collect EEG/EMG signals in freely moving awake animals to accurately distinguish wakefulness, NREM, and REM sleep stages.Technology Platform: Based on electrophysiology, continuously collect EEG/EMG signals in freely moving awake animals to accurately distinguish wakefulness, NREM, and REM sleep stages.
• Efficacy Evaluation: Cover standardized workflows such as multi‑model physiological sleep assessment, in vivo sleep EEG analysis, and sleep architecture analysis. Quantitatively evaluate drug effects on sleep onset, maintenance, and duration of each sleep stage, delivering reliable data support for sleep drug R&D.

Sources:
2025 China Sleep Health Survey Report
https://mp.weixin.qq.com/s/R0Vo6Cf3G_cgwzMCjMl1Yw
https://mp.weixin.qq.com/s/6duv-TmrSggkFmL7ip4gaQ?scene=1&click_id=49
Kylin Lab
CNSxplore | Pioneering CNS Drug Discovery--In vitro and in vivo, Beyond limits
Kylin Lab is a preclinical CRO company specializing in central nervous system (CNS) diseases, dedicated to offering one stop solutions for CNS drug discovery. With a portfolio of fully-validated cellular and animal disease models, combined with comprehensive research and analytical capabilities, we empower clients to accelerate the development of innovative therapies and reduce clinical trial risks.
Kylin Lab boasts an experienced technical team well-versed in international regulations, along with high-standard experimental platforms. Our core technologies include AI-driven phenotypic screening, humanized stem cells and organoids, electrophysiology, high-throughput electroencephalography (EEG), histology, and molecular biology. Our expertise spans a broad range of areas including Alzheimer’s disease, Parkinson’s disease, depression, schizophrenia, spinal muscular atrophy (SMA), pain, and stroke etc.
Since its establishment, Kylin Lab has adhered to the philosophy of “Pioneering R&D, Leading in technology Quality-first, Customer-centric.” We have served hundreds of clients, successfully completed numerous thematic research projects and IND submissions, and established a high-quality, stable, and forward-looking efficacy evaluation system.